|
In fact, one could argue that the history, struggle, and achievement that is threaded through the development of understanding matter at the atomic level is the quintessential story of the scientific method. 
This gradual, logical progression, where atomic structure evolved from being a simple, philosophical idea, through to the ultra-sophisticated world of the Higgs boson particle discovered in the early part of the 21 st century, represents a wonderful example of the evolution of a scientific idea, and the application of the scientific process. 
(You can read more about this is in our modules Early Ideas about Matter: From Democritus to Dalton and Atomic Theory I: The Early Days.) Despite the slow pace, it is crucial to understand that the process was a methodical one as each scientist built upon earlier ideas. 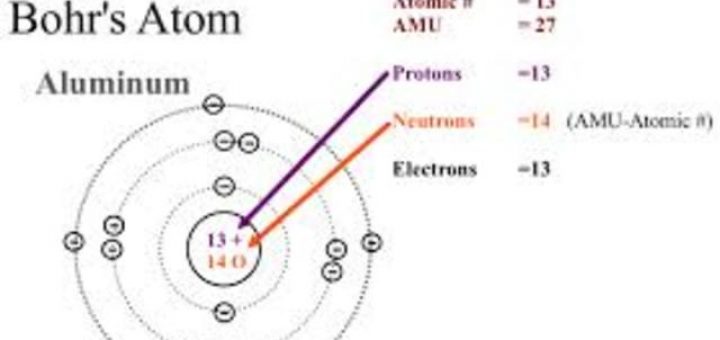
Starting with the ancient Greeks, and moving through to the beginning of the 19th century, the story unfolds relatively slowly. For example, giving 15.0 eV to an electron in the ground state of hydrogen strips it from the atom and leaves it with 1.4 eV of kinetic energy.The earliest ideas about matter at the atomic level were built over many centuries. Given more energy, the electron becomes unbound with some kinetic energy. Thus, 13.6 eV is needed to ionize hydrogen (to go from –13.6 eV to 0, or unbound), an experimentally verified number. N i n i size 12, and the electric potential energy thus becomes zero. Thus, for the Balmer series, n f = 2 n f = 2 and The constant n i n i is a positive integer, but it must be greater than n f n f. There are apparently an unlimited number of series, although they lie progressively farther into the infrared and become difficult to observe as n f n f increases. The Paschen series and all the rest are entirely IR. The Lyman series is entirely in the UV, while part of the Balmer series is visible with the remainder UV. For the Lyman series, n f = 1 n f = 1 for the Balmer series, n f = 2 n f = 2 for the Paschen series, n f = 3 n f = 3 and so on. The constant n f n f is a positive integer associated with a specific series. The observed hydrogen-spectrum wavelengths can be calculated using the following formula: (See Figure 30.16.) These series are named after early researchers who studied them in particular depth. The hydrogen spectrum had been observed in the infrared (IR), visible, and ultraviolet (UV), and several series of spectral lines had been observed. As you might expect, the simplest atom-hydrogen, with its single electron-has a relatively simple spectrum. In some cases, it had been possible to devise formulas that described the emission spectra. (credit for (b): Yttrium91, Wikimedia Commons) The line spectrum for each element is unique, providing a powerful and much used analytical tool, and many line spectra were well known for many years before they could be explained with physics. The discrete lines imply quantized energy states for the atoms that produce them. Part (b) shows the emission line spectrum for iron. (It was a running joke that any theory of atomic and molecular spectra could be destroyed by throwing a book of data at it, so complex were the spectra.) Following Einstein’s proposal of photons with quantized energies directly proportional to their wavelengths, it became even more evident that electrons in atoms can exist only in discrete orbits.įigure 30.15 Part (a) shows, from left to right, a discharge tube, slit, and diffraction grating producing a line spectrum. But, in spite of years of efforts by many great minds, no one had a workable theory. (See Figure 30.15.) Maxwell and others had realized that there must be a connection between the spectrum of an atom and its structure, something like the resonant frequencies of musical instruments. Atomic and molecular emission and absorption spectra have been known for over a century to be discrete (or quantized). (credit: Unknown Author, via Wikimedia Commons) Mysteries of Atomic SpectraĪs noted in Quantization of Energy, the energies of some small systems are quantized. His many contributions to the development of atomic physics and quantum mechanics, his personal influence on many students and colleagues, and his personal integrity, especially in the face of Nazi oppression, earned him a prominent place in history. Figure 30.14 Niels Bohr, Danish physicist, used the planetary model of the atom to explain the atomic spectrum and size of the hydrogen atom.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
